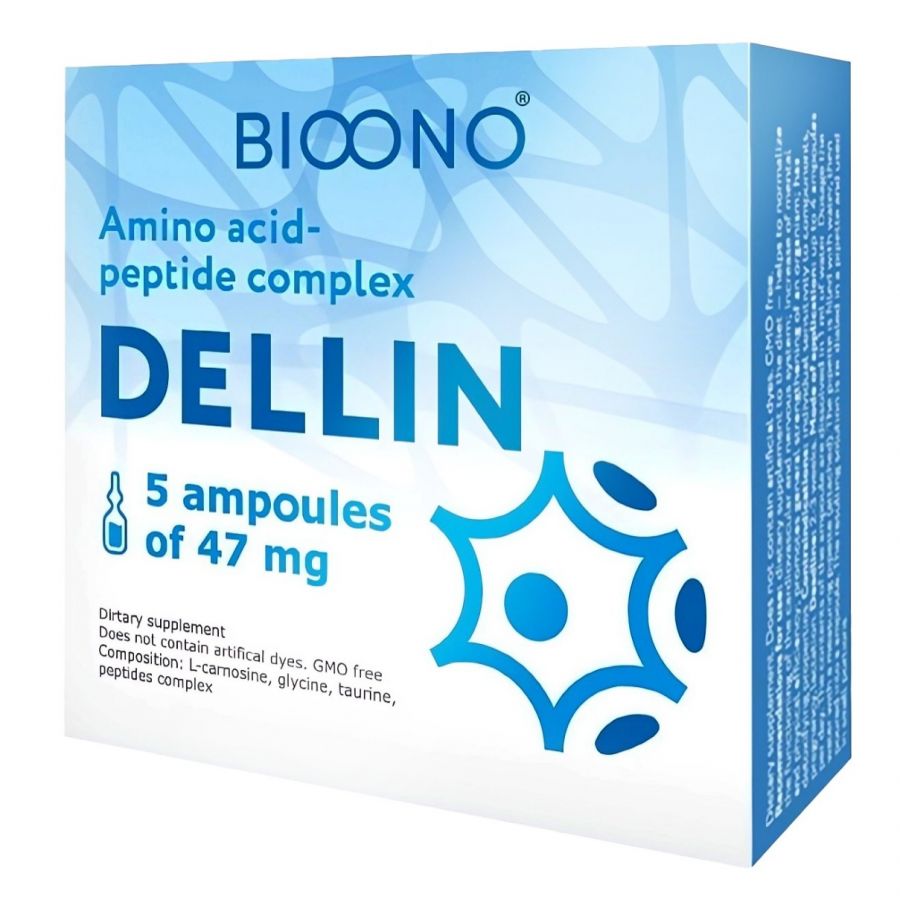
Dellin - Amino-acid Peptide Complex (DSIP included) - Dietary supplement
DELLIN - DSIP lyophilized powder for making drops in ampoules
Indications for the use of the DELLIN supplement:
- In pediatric neurology and psychiatry
- In general neurology and psychiatry:
- Complex therapy of epilepsy
- In drug practice:
*All results are individual and depend on your state of health
*Dellin is not a drug

- Buy 5 for €140.00 each and save 7%
- Buy 10 for €130.00 each and save 13%
Active ingredient: 1 ampoule contains delta-sleep peptide (0.0003 g) 0.3 mg; (buy dsip peptide)
Bioregulator DELLIN - is produced in the form of ampoules with odorless lyophilized powder. It is highly soluble in water. The ampoule contains 0.3 mg of the delta-sleep-inducing peptide DSIP and the rest is a specially selected mixture of glycine, turine, L-carnosine and pantel (an extract from reindeer antlers).
Indications for the use of the DELLIN supplement:
- In pediatric neurology and psychiatry
- In general neurology and psychiatry:
- Complex therapy of epilepsy
- In drug practice:
- Peptide Therapy for Sleep
- Neuropeptide
Courses for children over 14 years old and adults:
Standard or planned therapy of the pathological process - take 1 ampoule from 10 to 30 days, if necessary, after a 10-15 day pause, repeat the course of 1 ampoule daily for 10-15 days;
Early period of rehabilitation of an acute, severe, chronic pathological process - take 2 ampoules for 15-20 days and repeat the course after a 10-day break. Further use is subject to the current state;
Early rehabilitation period for an acute but milder pathological process - take 2 ampoules for 5 days, and then 1 ampule for 15-20 days and after a 15-day pause, repeat 1 ampule daily for 10-15 days;
Complex therapy of an acute pathological process - take 2 ampoules for 10-15 days) and after a 15-day pause, repeat 1 ampoule daily for 10 days;
If the patient is undergoing resuscitation or intensive therapy, it is possible to use from 3 to 5 ampoules of the DELLIN Bioregulator per day, for 2-5 days, and then continue therapy according to the protocol for an acute, severe, chronic pathological process.
Content: Carnosine, a dipeptide consisting of two amino acids (alanine and histidine), is a natural component of human tissue: Glycine (aminoacetic acid) - refers to the nonessential amino acids, is a part of many proteins and biologically active compounds, is easily absorbed by the body. Taurine is an essential sulfoamino acid that has been found in almost all mammalian species.
Peptide complex - including delta-peptide with the last Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu, amino acids, growth factor.
Recommended daily dosage: 1-4 ampoules a day during 2-4 weeks.
- Composition in 1 ampoule (47 mg):
- Carnosine – 20 mg
- Glycine – 20 mg
- Taurine – 5 mg
- Peptide complex – 2 mg
Does not contain: artificial flavors or sweeteners, preservatives, sugar, starch, milk, lactose, soy, eggs, glutamate, wheat, yeast, magnesium stearate. Without GMO.
Store in a dry place at temperatures between 5 0 С and 25 0 C, away from direct sunlight.
- Contents: 5 ampoules
- Contents: 235 mg
DELLIN CONTAINS:
The effective results achieved with DELLIN are a consequence of the unique combination of ingredients included in its composition:
Delta sleep peptide DSIP as the main active component Dellin is a natural neuromodulatory peptide that is produced in the body and has broad pharmacological activity. This peptide exhibits a pronounced stress-protective and adaptogenic effect, increases the body’s resistance to the effects of various unfavorable stress factors in pathological conditions due to diseases of various etiologies. The delta sleep peptide prevents the occurrence or limits the severity of stress-induced pathological processes in the body; its modulating effect does not manifest itself in the normal physiological state of the body. Delta sleep peptide has antidepressant and anticonvulsant effects, normalizes sleep, exhibits antitoxic properties, increases mental and physical performance, and limits autonomic disorders. This neuropeptide increases the electrical stability of the heart and increases the fibrillation threshold, limiting cardiovascular disorders under stress. Delta sleep peptide reduces the primary pathological craving for alcohol and eliminates symptoms of alcohol withdrawal.
DELLIN CONTAINS:
Dellin is a unique and powerful mixture of biologically active substances that guarantees its therapeutic effectiveness. This complex includes nerve growth factors and various peptides, including delta sleep peptide.
Dellin's complex composition also includes a wide range of free amino acids, each of which contributes to the complex effect of the drug on physiological processes:
The components of this complex include free amino acids with different Rf values: aspartic acid (0.51 ± 0.015), serine (0.46 ± 0.071), glycine (0.41 ± 0.018), histidine (0.13 ± 0.030), arginine ( 0.19 ± 0.025), tyrosine (0.43 ± 0.020), alanine (0.38 ± 0.043), threonine (0.31 ± 0.011), valine (0.56 ± 0.014), methionine (0.27 ± 0.037 ), leucine (0.64 ± 0.075), phenylalanine (0.81 ± 0.031) and lysine (0.09 ± 0.025).
It also contains minerals, presented both in the form of ionic and chelate complexes. Macroelements include iron, calcium, magnesium, sodium, phosphorus and potassium. The microelements include manganese, selenium, cobalt, copper, zinc and iodine.
The lipid fraction includes phospholipids, cerebroside, colaminkephalin, lecithin and other components.
The peptides NGF-1 and NGF-3, which belong to the neurotrophin family, deserve special attention. These small secreted proteins are important for maintaining the viability and activity of neurons, promoting their development. They are essential for the survival of sympathetic and sensory neurons by preventing their apoptosis. Nerve growth factors stimulate the growth of axons, promoting their branching and elongation. NGF-1 interacts with at least two classes of receptors, LNGFR and TrkA, associated with neurodegenerative diseases.
NGF is key to maintaining homeostasis in the body by circulating throughout its systems. It prevents or reduces neuronal degeneration in neurodegenerative diseases, which has been proven in animal experiments and clinical trials in humans. NGF is also involved in suppressing inflammatory processes and restoring myelin.
NGF (nerve growth factor) plays a critical role in preventing or reducing neuronal degeneration in animals suffering from neurodegenerative diseases. These positive research findings in animal models prompted a series of clinical trials in humans. There is an increase in NGF expression in inflammatory settings, where it effectively reduces inflammation. NGF is also active in remyelination processes.
DSIP (delta sleep-inducing peptide) functions to stabilize neural structures and interneuron connections during the delta phase of sleep. It stimulates the synthesis of neurotrophins, restores active synaptic connections and prevents excessive excitation of neurons during adaptive reactions, protecting them from self-destruction through the mechanism of excitotoxicity (“Exito Cyto Toxicity”).
Carnosine , chemically known as beta-alanyl-L-histidine, is a dipeptide formed by combining the amino acid residues beta-alanine and histidine. This dipeptide is highly concentrated in muscle tissue and the brain, highlighting its biological significance in these areas.
Main properties of carnosine:
Carnosine, a biologically active beta-alanyl-L-histidine dipeptide, has many properties that make it an outstanding antioxidant:
-
Natural Origin and Synthesis in the Body: Carnosine is naturally synthesized in the human body, especially in muscle and nerve tissues.
-
Penetration Through the Blood-Brain Barrier: It is able to penetrate the brain through the blood-brain barrier, making it particularly effective in protecting the nervous system.
-
High Bioavailability and Membrane Stabilizing Action: Carnosine is easily absorbed by the body and has cell membrane stabilizing properties.
-
Hydrophilic Low Molecular Antioxidant: It is a direct-acting hydrophilic antioxidant, which increases its effectiveness in combating oxidative stress.
-
No Side Effects or Addictiveness: Carnosine has no side effects or signs of addiction, making it safe for long-term use.
-
No Risk of Overdose: Carnosine does not accumulate in the body, since its excess is broken down by the enzyme carnosinase into its constituent amino acids, which are easily removed from the body.
These properties make carnosine one of the most promising and safe antioxidants widely studied in modern science.
Initially, the positive biological effects of carnosine were associated mainly with its pH-buffering properties. These properties allow carnosine to maintain stable acidity levels in cells, especially muscle tissue, which prevents damage caused by acidosis.
However, subsequent studies revealed that carnosine also has direct antioxidant effects. This discovery expanded the understanding of its functions, making it not only a buffer for protons, but also a buffer for variable valence metals and reactive oxygen species. Carnosine, acting as an antioxidant, neutralizes free radicals and protects cells from oxidative stress caused by external and internal factors.
In addition, its antiglycating and anticrosslinking properties were discovered. Glycation is the process by which sugars combine nonenzymatically with proteins, lipids, or nucleic acids, which can lead to the formation of harmful advanced glycation end products (AGEs). Carnosine effectively prevents this process, thereby protecting cellular structures from damage and aging. The anti-crosslinking effect of carnosine helps prevent the formation of pathological bonds between molecules, which also helps slow down the aging process and improve cellular functioning.
Thus, carnosine acts not only as a powerful antioxidant, but also as an important protector of cells from many processes that contribute to their aging and degradation.
The use of carnosine in the treatment of neuropsychiatric and mental disorders is due to its antioxidant properties and ability to penetrate the blood-brain barrier:
-
Combat Oxidative Stress: Oxidative stress plays a key role in the development of neurological diseases such as Parkinson's and Alzheimer's diseases, as well as stroke. Carnosine, having antioxidant properties, can neutralize free radicals, thereby protecting nerve cells from damage.
-
Protection of Nerve Cells: The nervous system is especially vulnerable to free radical oxidation due to high levels of metabolic processes, oxygen consumption, the presence of lipids with polyunsaturated fatty acids and other factors. Carnosine helps in protecting nerve cells from these harmful factors.
-
Uses in Psychiatric Disorders: Carnosine may have beneficial effects in schizophrenia, depression, and addictive disorders such as alcoholism, as these conditions are often associated with increased oxidative stress and metabolic disturbances in the nervous system.
-
Crossing the Blood-Brain Barrier: One of the key benefits of carnosine is its ability to cross the blood-brain barrier, allowing it to have a direct effect on brain cells.
Thus, due to its unique properties, carnosine represents an important potential for the treatment and prevention of various neuropsychiatric and mental diseases.
Positive results were obtained by adding carnosine to the basic therapy of patients with chronic dyscirculatory encephalopathy. This treatment led to increased resistance of blood plasma lipoproteins to Fe2+-induced oxidation, stabilization of erythrocytes with respect to acid hemolysis, intensification of the respiratory burst of leukocytes and strengthening of the body's endogenous antioxidant defense, and improvement of the cognitive functions of the brain of patients. That is, carnosine had antioxidant, membrane-stabilizing and immunomodulatory effects in this pathology.
Research on the use of carnosine in the treatment of Parkinson's disease has shown encouraging results:
-
Improvement in Clinical Condition : When carnosine was added to standard therapy for Parkinson's disease for 30 days, there was a significant improvement in the overall clinical condition of patients.
-
Reducing the Toxic Effects of Traditional Therapy : Carnosine has helped reduce the negative side effects of anti-Parkinson's drugs.
-
Reduction in Neurological Symptoms: Carnosine use statistically significantly reduced the occurrence of neurological symptoms such as incoordination.
-
Activation of Antioxidant Enzyme Superoxide Dismutase : A positive correlation has been observed between increased superoxide dismutase activity in red blood cells and a decrease in neurological symptoms.
-
Reducing the Level of Hydroperoxides in Lipoproteins : The addition of carnosine to treatment led to a decrease in the level of hydroperoxides in plasma lipoproteins.
-
Increased Resistance of Lipoproteins to Oxidation : Carnosine significantly increased the resistance of low and very low density lipoproteins to oxidation caused by Fe2+.
-
Reduction in the Amount of Oxidized Proteins in Blood Plasma : A decrease in the level of oxidized proteins in blood plasma has been recorded in patients receiving carnosine.
These data indicate that carnosine may be an effective adjunct to conventional therapy for Parkinson's disease, improving both clinical parameters and the body's overall antioxidant status. This suggests a potential role for carnosine as an adjuvant in the treatment of neurological disorders.
Carnosine is also helpful in improving brain function in autism. In one double-blind, placebo-controlled study of 301 children with autism, carnosine was found to improve expressive and receptive vocabulary and cause objective improvement on an autism rating scale.
Carnosine has potential as an overall anti-aging agent due to a number of its unique properties:
-
Slowing the Aging Process: Clinical studies indicate the potential for slowing the aging process due to carnosine's properties to prevent oxidative damage and glycation. These processes are key in the mechanisms of aging at the cellular level.
-
Inhibition of Inflammatory Mediators: Carnosine has been shown to be effective in reducing the release of inflammatory mediators, including various cytokines. This reduces asymptomatic inflammation, which is important not only for slowing down aging, but also for preventing chronic degenerative diseases.
-
Prevention of Chronic Diseases: Reducing inflammation helps prevent the development of diseases such as cardiovascular disease and diabetes, as well as neurodegenerative disorders including Parkinson's and Alzheimer's diseases.
-
Cognitive and Memory Support : Given carnosine's ability to cross the blood-brain barrier and influence brain processes, it may be effective in preventing cognitive and memory decline associated with age-related changes.
Thus, carnosine may be a valuable addition to anti-aging strategies and the prevention of chronic diseases associated with aging due to its antioxidant, anti-inflammatory and neuroprotective properties.
- European patent A61K45/00 indicates that the combination of carnosinase inhibitors with carnosine enhances the therapeutic effect (Babizhaev M., Meguro K.)
- US patent US2008/0171095 A1 shows that carnosine reduces the ischemic penumbra in ischemic stroke (Majid M., Krisanamurthy R.)
- Russian patent No. RF2353382 (09/12/2007) demonstrates increased effectiveness of therapy for Parkinson's disease when combining carnosine with classical therapy (Boldyrev A. A. et al.)
Glycine, being a neurotransmitter amino acid, plays an important role in the functioning of the brain and nervous system, and is also involved in various metabolic processes:
-
Dual Action on Neurons : Glycine has an inhibitory effect on neurons by decreasing the release of excitatory neurotransmitters such as glutamic acid and increasing the release of GABA (gamma-aminobutyric acid). It binds to glycine receptors (encoded by the GLRA1, GLRA2, GLRA3 and GLRB genes) in the brain and spinal cord.
-
Interaction with NMDA receptors : Glycine also binds to NMDA receptors, promoting the transmission of signals from excitatory neurotransmitters such as glutamate and aspartate.
-
Cell Bioenergetics Support : Glycine is involved in maintaining bioenergetic processes in the cell and is an antihypoxant, helping cells survive in conditions of oxygen deficiency.
-
Participation in Protein Synthesis : As a non-essential amino acid, glycine is part of polypeptide chains, forming the primary structure of proteins.
-
Role in the Synthesis of Important Biomolecules: Glycine takes part in the synthesis of purines, porphyrins, creatine and phospholipids important for cell membranes.
-
Glutathione Synthesis : Glycine is important for the synthesis of glutathione, a natural antioxidant and source of SH groups that protects the cell from free radicals.
-
Enhance Cell Compensatory Capabilities: Activation of glutathione helps increase the cells' ability to cope with oxidative stress.
-
Detoxification of Xenobiotics: Glycine is capable of direct non-specific conjugation with xenobiotics, forming less toxic metabolites, which helps detoxify substances harmful to the cell.
-
Use in Acute Stroke : As a detoxifier, glycine binds aldehydes and ketones, which are formed in large quantities during acute stroke, reducing their toxic effect.
Thus, glycine has a wide range of biological functions, from neurotransmitter activity to participation in metabolic processes and protection of cells from damage.
Taurine is a sulfonic acid formed in the body from the amino acid cysteine. Taurine is often called a sulfur-containing amino acid, and the molecule lacks a carboxyl group.
Recently, it has been established that in the brain taurine plays the role of a neurotransmitter amino acid that inhibits synaptic transmission and has anticonvulsant activity. Taurine helps improve energy processes, stimulates healing processes in dystrophic diseases and processes accompanied by significant metabolic disorders of eye tissue. There is evidence that taurine promotes the formation of new cells in the hippocampus, an area of the brain associated with memory. It also promotes brain regeneration in cases of closed head injuries.
Taurine, as an important neurotransmitter and neuromodulator, may have protective effects on neurons against glutamate-induced excitotoxicity, which is key to neuronal stability and brain function:
-
Reduced D-Aspartate Release: Taurine may reduce the release of D-aspartate, which is an analogue of L-glutamate, one of the main excitatory neurotransmitters in the CNS. This action may help reduce neuronal excitation and reduce the risk of excitotoxicity.
-
Protection against Glutamate Excitotoxicity: Taurine may protect neurons from glutamate-induced excitotoxicity by reducing intracellular calcium ion (Ca2+) levels. Glutamate excitotoxicity is associated with excess calcium ion entry into the cell, which can lead to neuronal damage and death.
-
Effect on Chloride Channels: Taurine may also affect the functioning of chloride channels by preventing cell membrane depolarization caused by glutamate. This prevents the activation of pathological cascades associated with excessive excitation of neurons.
-
Balance Between Excitatory and Inhibitory Systems: Taurine may play a role in establishing a balance between excitatory and inhibitory mechanisms in the brain. It is critical for maintaining normal brain function and preventing neurological disorders.
Being a structural analogue of the main inhibitory transmitter GABA, it interacts with GABAA receptors, activating them, but to a lesser extent than GABA itself. Among all GABAA receptors, taurine most strongly affects those containing the b2 subunit, localized in mammals in the dentate gyrus, substantia nigra, molecular layer of the cerebellum, medial nucleus of the thalamus, field CA3 of the hippocampus. The release of taurine from neurons also reduces cell swelling and thereby helps regulate osmosis in a state of excitotoxicity.
Free radical oxidation processes in the body are controlled by the antioxidant system. The leading role in maintaining the antioxidant status of the cell belongs to glutathione peroxidase and glutathione reductase. The main function of these enzymes is the reduction of hydroperoxides to alcohols. As the results of the study showed, with cerebral edema there is a decrease in glutathione content and the activity of glutathione peroxidase and glutathione reductase.
In animals and humans, glutathione is present in both oxidized (GSSG; about 10% of the total) and reduced (GSH) forms. The main antioxidant effect of glutathione is realized through its participation in the work of enzymatic antioxidants; being a substrate for glutathione peroxidases, it actually acts as a donor of hydrogen atoms for the reduction of H2O2 and lipid peroxides.
Due to the fact that a decrease in the level of glutathione and antioxidant enzymes is one of the leading factors in the development of various pathological processes, substances that increase glutathione content and activate glutathione-dependent reactions are of great interest. The amino acid taurine acts as such a substance. There is convincing evidence of the role of taurine as an active osmoregulator, which is especially important for brain neurons. A correlation has been shown between the content of water and taurine in brain tissue. In hepatic encephalopathy, a decrease in taurine content in the central nervous system may be one of the causes of cerebral edema. It is also involved as a neuromodulator in the control of respiratory function, especially during acute hypoxia.
The results of studies related to the administration of taurine demonstrate its significant effect on biochemical processes in the brain, especially in patients with cerebral edema:
-
Reducing Lipid Peroxidation: Taurine promotes the elimination of lipid peroxidation products. This process, characteristic of oxidative stress, is one of the main causes of damage to cell membranes and can contribute to the development of neurological disorders.
-
Normalizes Oxidative Modification of Proteins in Mitochondria: Taurine also has a positive effect on mitochondrial proteins, helping to reduce or normalize their oxidative modification. Mitochondria play a key role in cellular energy metabolism, and their function is critical to the health of the nervous system.
-
Effect on Cerebral Edema: Since cerebral edema is often accompanied by increased oxidative stress and impaired mitochondrial function, taurine administration may be beneficial in reducing these pathological changes. Thus, taurine may help improve the condition of individuals with cerebral edema.
These results highlight the potential role of taurine as a neuroprotective agent that may be useful in treating conditions associated with increased oxidative stress and impaired mitochondrial function, such as cerebral edema.
Research conducted in the 1970s and subsequent clinical trials have identified taurine's potential role in reducing the frequency and intensity of epileptic seizures.
Key points from these studies include:
-
Animal Experiments: In the 1970s, there were approximately 20 reports showing that taurine could attenuate epileptic seizures in various animal models.
-
Transition to Clinical Research: The positive results of these experiments provided the impetus for conducting clinical trials in humans. These trials were aimed at studying the effect of taurine on epilepsy in humans.
-
Effect of Taurine on Neuronal Excitation: Clinical evidence has shown that reducing taurine levels in the brain may increase overall neuronal excitation, which in turn may contribute to the occurrence of epileptic seizures.
-
Publications and Specialist Interest: Following the publications of French and Finnish scientists on this topic, interest in the role of taurine in the treatment of epilepsy has increased significantly. One of the important works in this area is the article by SS Ojaa and P. Saransaari, published in the journal Epilepsy Research in 2013, where the authors summarized the accumulated knowledge on this topic.
These data highlight the importance of taurine in the context of neurobiology and treatment of epilepsy, supporting the need for further research to better understand its mechanisms of action and potential therapeutic use.